 For any given isotope, the sum of the numbers of protons and neutrons in the nucleus is called the mass number. These different types of helium atoms have different masses (3 or 4 atomic mass units), and they are called isotopes. For example, stable helium atoms exist that contain either one or two neutrons, but both atoms have two protons. 
The element helium will always have two protons in its nucleus.Ītoms of the same element can, however, have differing numbers of neutrons in their nucleus. For example, the element hydrogen (the lightest element) will always have one proton in its nucleus. The atomic number of an element defines the element's identity and signifies the number of protons in the nucleus of one atom. 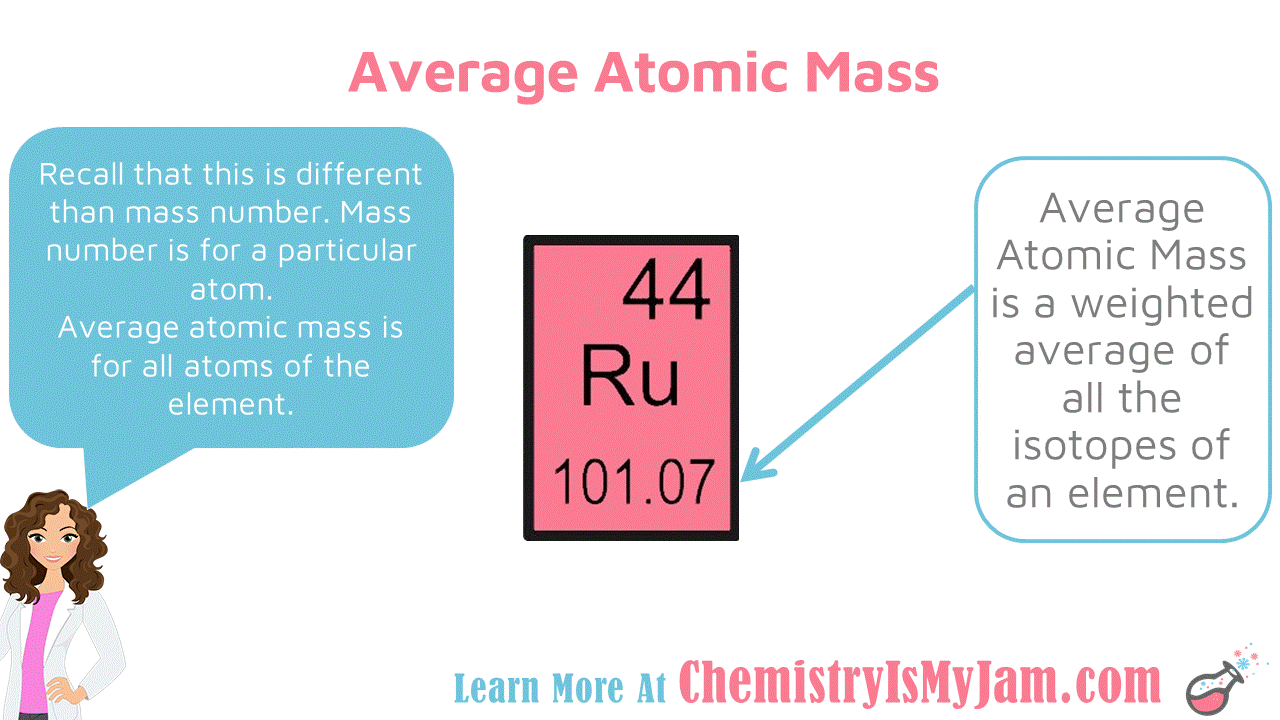
mass numberThe total number of protons and neutrons in an atomic nucleus.average atomic massThe mass calculated by summing the masses of an element's isotopes, each multiplied by its natural abundance on Earth.natural abundanceThe abundance of a particular isotope naturally found on the planet.When doing any mass calculations involving elements or compounds, always use average atomic mass, which can be found on the periodic table.The average atomic mass for an element is calculated by summing the masses of the element's isotopes, each multiplied by its natural abundance on Earth. 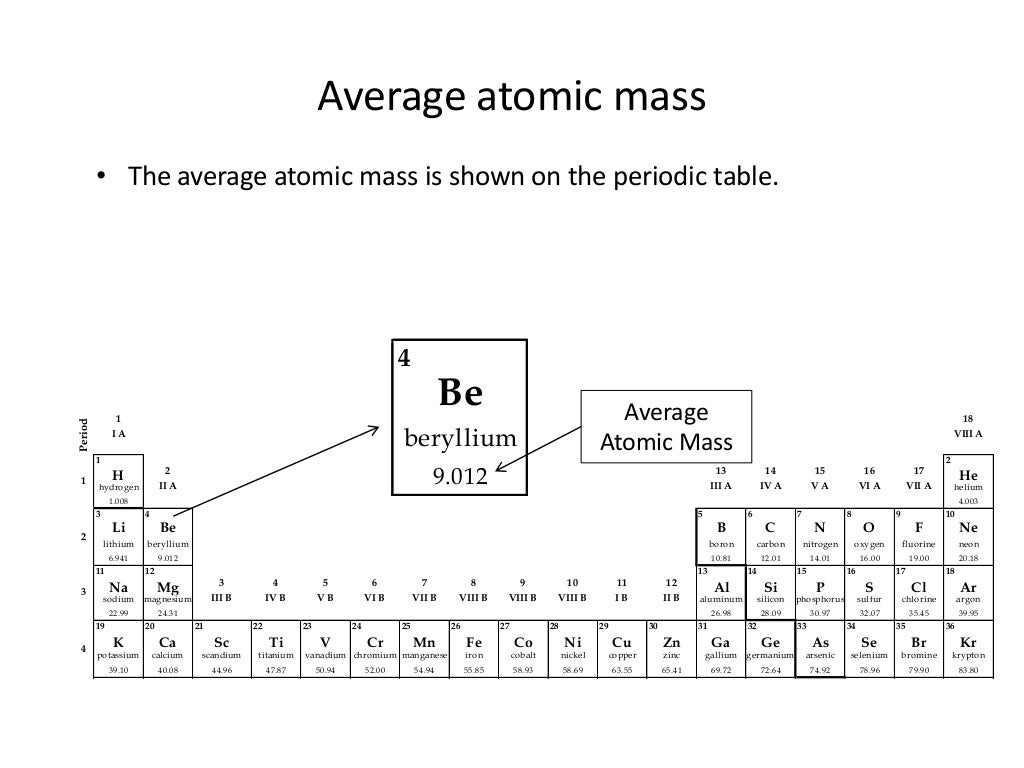
The versions of an element with different neutrons have different masses and are called isotopes. An element can have differing numbers of neutrons in its nucleus, but it always has the same number of protons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
